
It's important to know the facts when you are treating patients for Postural Orthostatic Tachycardia Syndrome. Here's what we know:
The WHO Recommendation
The World Health Organization has outlined the best methods t0 prevent dehydration in individuals with water loss due to chronic illnesses or acute illness like diarrhea. Fairly recently the WHO outlined a new formula that has revolutionized the efficiency of rehydration. Some have hailed it as the most significant discovery in modern medicine.
In the WHO's discovery there is a very specific recipe for oral rehydration. You can read the white papers on ORS (oral rehydration salts) for dehydration here.
NormaLyte follows the WHO recommendations exactly.
NormaLyte is Clinically Studied

In a clinical study, Oral Rehydration Salts like NormaLyte were proven to be more effective than saline IV alone.
Read the study details to determine if NormaLyte is a good solution for your patients, and view the charts below from the study:
Figure 1.
The response of a representative patient with OI to the imposition of a controlled orthostatic challenge using LBNP. The top panel shows that in the absence of supplemental fluid administration (No Fluid), presyncope occurred during exposure to −30 mmHg negative pressure. Following IV saline (Saline, middle panel), and after ingestion of oral rehydration solution (ORS, lower panel), enhanced orthostatic tolerance was achieved as presyncope occurred during exposure to −60 mmHg negative pressure.

Figure 2.
Changes in cardiac output (CO in L/min) in control (top panel) and POTS patients with OI (bottom panel) during imposition of a controlled orthostatic challenge using LBNP following no treatment (No TX – white bars), IV sal ine (Saline – gray bars) and ingestion of oral rehydration solution (ORS – black bars). Treatment effects for controls were not significantly different. For POTS, CO was significantly higher following ORS, compared with no treatment (* = No Tx, p<0.05), both at baseline and −45mmHg and saline had no significant effect.
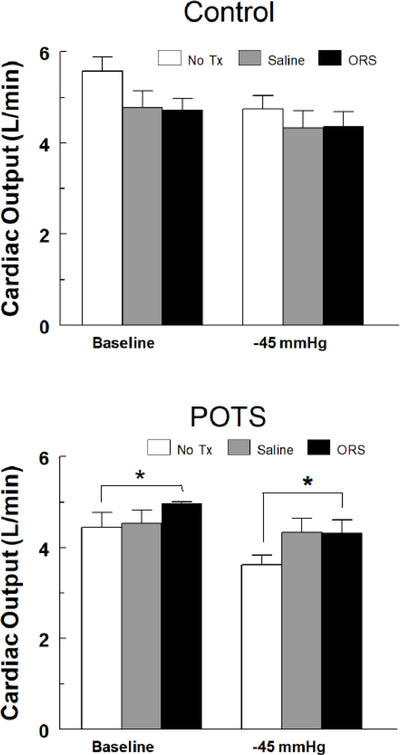
Figure 3.
Changes in normalized cerebral blood flow velocity (normalizedCBFV) in control (top panel) and POTS patients with OI (bottom panel) during imposition of a controlled orthostatic challenge using LBNP following no treatment (No TX – white bars), IV saline (Saline – gray bars) and ingestion of oral rehydration solution (ORS – black bars). Because there was no pressure effect, the comparisons reflect the combined effect (i.e., the weighted average) of the CBVv in the ORS group vs. No Tx. In controls, normalized CBFv was not significantly reduced at −45 mmHg, nor did fluid administration result in any significant change. In POTS, CBFv following ORS was significantly higher than No TX (ǂ = p<0.0005), which was independent of pressure.

Figure 4.
Changes in normalized orthostatic index (% of that measured without treatment in control (black bars) and POTS patients with OI (gray bars) measured following no treatment (Untreated), intravenous saline (IV Saline) and ingestion of oral rehydration solution (ORS). Neither saline nor ORS increased orthostatic tolerance in untreated controls (p=0.46, N=15); both I.V. saline and ORS significantly improved orthostatic tolerance (* = p<0.05 and ** = p<0.001, respectively, N=10) in POTS subjects.









